Adaptation and validation of the Spanish version of the thyroid-cancer-specific Thyca-QoL scale: A cross-sectional study
DOI:
https://doi.org/10.30944/20117582.2177Palabras clave:
neoplasias tiroideas, calidad de vida, comparación transcultural, estudio de validación, psicometríaResumen
Introduction. Thyca-QoL is a specific instrument to assess QoL in thyroid cancer patients, but it is not validated in Spanish language. The aim was to assess the psychometric properties of the Thyca-QoL.
Methods. This is a prospective cross-sectional study. The Thyca-QoL was translated and adapted to Spanish language. A psychometric validation using an exploratory principal axis factor analysis and confirmatory analysis, concurrent validation compared with EORTC QLQ-C30 and a test-retest reliability assessment was done.
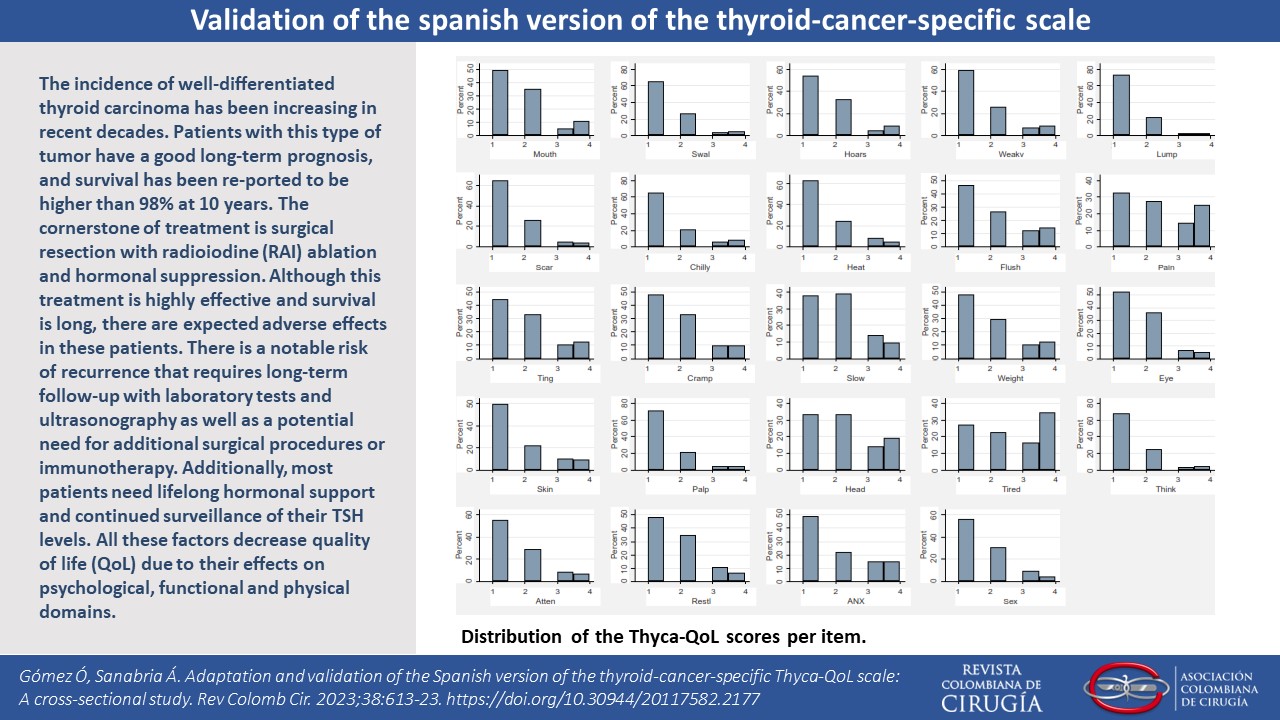
Results. A total of 296 patients were included. Exploratory factor analysis showed a seven-factor solution with good diagnostic tests results. Cronbach´s alpha for the global scale was 0.86. The comparison between the ThycaQoL and the EORTC QLQ-C30 demonstrated a high correlation (rho= 0.75) and coefficient for test-retest was 0.87.
Discussion. The validation process followed all the methodological steps necessary to guarantee the performance of the instrument. The measurements of the internal validity, reliability and reproducibility reached similar results as the original validation. The factor analysis showed a solution with seven factors that resembles the original results. Reproducibility was high for voice, sympathetic, sex, and chilliness domains and moderate for the others and the instrument had the ability to discriminate between clinical conditions.
Conclusion. Thyca-QoL is a reliable and objective instrument to be used in clinical practice and for research objectives in Spanish speaking patients.
Descargas
Referencias bibliográficas
Davies L, Morris L, Hankey B. Increases in thyroid cancer incidence and mortality. JAMA. 2017;318:389-390. https://doi.org/10.1001/jama.2017.7906
Sánchez G, Díaz M, Ángel I, Ariza A. Carcinoma de tiroides: Descripción de 634 pacientes atendidos en el Hospital Universitario San Ignacio, Bogotá, D.C., Colombia. Rev Colomb Cir. 2022;37: 588-596. https://doi.org/10.30944/20117582.2179
Vargas-Pinto S, Romero Arenas MA. Lobectomy compared to total thyroidectomy for low-risk papillary thyroid cancer: A systematic review. J Surg Res. 2019;242:244- 251. https://doi.org/10.1016/j.jss.2019.04.036
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26:1-133. https://doi.org/10.1089/thy.2015.0020
Husson O, Haak HR, Mols F, Nieuwenhuijzen GA, Nieuwlaat WA, Reemst PH, et al. Development of a disease-specific health-related quality of life questionnaire (THYCA-QoL) for thyroid cancer survivors. Acta Oncol. 2013;52:447-54. https://doi.org/10.3109/0284186X.2012.718445
Vissers PAJ, Thong MSY, Pouwer F, Zanders MMJ, Coebergh JWW, van de Poll-Franse LV. The impact of comorbidity on Health-Related Quality of Life among cancer survivors: analyses of data from the PROFILES registry. J Cancer Surviv. 2013;7:602-13. https://doi.org/10.1007/s11764-013-0299-1
Singer S, Lincke T, Gamper E, Bhaskaran K, Schreiber S, Hinz A, et al. Quality of life in patients with thyroid cancer compared with the general population. Thyroid. 2012;22:117-24. https://doi.org/10.1089/thy.2011.0139
Tan LGL, Nan L, Thumboo J, Sundram F, Tan LKS. Health-related quality of life in thyroid cancer survivors. Laryngoscope. 2007;117:507-10. https://doi.org/10.1097/MLG.0b013e31802e3739
Sawka AM, Naeem A, Jones J, Lowe J, Segal P, Goguen J, et al. Persistent posttreatment fatigue in thyroid cancer survivors: a scoping review. Endocrinol Metab Clin North Am. 2014;43:475-94. https://doi.org/10.1016/j.ecl.2014.02.007
Husson O, Haak HR, Buffart LM, Nieuwlaat WA, Oranje WA, Mols F, et al. Health-related quality of life and disease specific symptoms in long-term thyroid cancer survivors: a study from the population-based PROFILES registry. Acta Oncol. 2013;52:249-58. https://doi.org/10.3109/0284186X.2012.741326
Carrillo JF, Ortiz-Toledo MA, Salido-Noriega Z, Romero-Ventura NB, Ochoa-Carrillo FJ, Oñate-Ocaña LF. Validation of the Mexican Spanish version of the EORTC QLQ-H&N35 instrument to measure health-related quality of life in patients with head and neck cancers. Ann Surg Oncol. 2013;20:1417-26. https://doi.org/10.1245/s10434-012-2712-9
Lee JI, Kim SH, Tan AH, Kim HK, Jang HW, Hur KY, et al. Decreased health-related quality of life in disease-free survivors of differentiated thyroid cancer in Korea. Health Qual Life Outcomes. 2010;8:101. https://doi.org/10.1186/1477-7525-8-101
Husson O, Haak HR, Oranje WA, Mols F, Reemst PHM, van de Poll-Franse LV. Health-related quality of life among thyroid cancer survivors: a systematic review. Clin Endocrinol (Oxf). 2011;75:544-54. https://doi.org/10.1111/j.1365-2265.2011.04114.x
Novoa-Gomez MM, Vargas-Gutierrez RM, Obispo-Castellanos SA, Pertuz-Vergara M, Rivera-Padilla YK. Psychological well-being and quality of life in patients treated for thyroid cancer after surgery Terapia Psicologica. 2010;28:69-84.
Botella-Carretero JI, Galan JM, Caballero C, Sancho J, Escobar-Morreale HF. Quality of life and psychometric functionality in patients with differentiated thyroid carcinoma. Endocr Relat Cancer. 2003;10:601-10. https://doi.org/10.1677/erc.0.0100601
Vega-Vazquez MA, Gonzalez-Rodriguez L, Santiago-Rodriguez EJ, Garces-Dominguez A, Shum LM, Tirado-Gomez M, et al. Quality of life-in patients with differentiated thyroid cancer at the general endocrinology clinics of the University Hospital of Puerto Rico. Bol Asoc Med P R. 2015;107:25-31.
Patrick DL, Burke LB, Gwaltney CJ, Leidy NK, Martin ML, Molsen E, et al. Content validity--establishing and reporting the evidence in newly developed patient-reported outcomes (PRO) instruments for medical product evaluation: ISPOR PRO good research practices task force report: part 1--eliciting concepts for a new PRO instrument. Value Health. 2011;14:967-77. https://doi.org/10.1016/j.jval.2011.06.014
Rothman M, Burke L, Erickson P, Leidy NK, Patrick DL, Petrie CD. Use of existing patient-reported outcome (PRO) instruments and their modification: the ISPOR good research practices for evaluating and documenting content validity for the use of existing instruments and their modification PRO Task Force Report. Value Health. 2009;12:1075-83. https://doi.org/10.1111/j.1524-4733.2009.00603.x
Wild D, Grove A, Martin M, Eremenco S, McElroy S, Verjee-Lorenz A, et al. Principles of Good Practice for the translation and cultural adaptation process for patient-reported outcomes (PRO) measures: Report of the ISPOR Task Force for translation and cultural adaptation. Value Health. 2005;8:94-104. https://doi.org/10.1111/j.1524-4733.2005.04054.x
Bonnet DG. Sample size requirements for testing and estimating coefficient alpha. Journal of Educational and Behavioral Statistics. 2002;27:335-40. https://doi.org/10.3102/10769986027004335
Applewhite MK, James BC, Kaplan SP, Angelos P, Kaplan EL, Grogan RH, et al. Quality of life in thyroid cancer is similar to that of other cancers with worse survival. World J Surg. 2016;40:551-61. https://doi.org/10.1007/s00268-015-3300-5
Jeong Y, Choi J, Ahn AL, Oh EJ, Oh HK, Cho DY, et al. Validation of the Korean version of the thyroid cancer-specific quality of life questionnaire. Ann Surg Treat Res. 2015;89:287-294. https://doi.org/10.4174/astr.2015.89.6.287
Descargas
Publicado
Cómo citar
Número
Sección
Licencia
Derechos de autor 2023 Revista Colombiana de Cirugía

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-SinDerivadas 4.0.
Todos los textos incluidos en la Revista Colombiana de Cirugía están protegidos por derechos de autor. Las opiniones expresadas en los artículos firmados son las de los autores y no coinciden necesariamente con las de los directores o los editores de la Revista Colombiana de Cirugía. Las sugerencias diagnósticas o terapéuticas como elección de productos, dosificación y métodos de empleo corresponden a la experiencia y al criterio de los autores.

















.jpg)