Inestabilidad microsatelital y cáncer gástrico
DOI:
https://doi.org/10.30944/20117582.658Palabras clave:
cáncer, gástrico, inestabilidad microsatelital, inmunoterapia, tratamientoResumen
La inestabilidad microsatelital es causada por una alteración de los sistemas de reparación de apareamiento
incorrecto, que puede afectar los microsatélites dentro de todo el genoma humano, produciendo errores en su replicación. Los estudios publicados, principalmente en la literatura inglesa, han encontrado que algunos tumores, como los gástricos, pueden expresar inestabilidad microsatelital.
En la siguiente revisión de tema, se presenta una descripción de los sistemas de reparación de apareamientos incorrectos y su relación con la presencia de inestabilidad microsatelital en los tumores gástricos, así como su posible utilidad clínica, como factor asociado en la respuesta al tratamiento con inmunoterapia en los pacientes con dicha patología.
Descargas
Referencias bibliográficas
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. https://doi.org/10.3322/caac.21492
Organization WH. Globocan 2018 Lyon, France: International Agency for Research on Cancer; 2018 [cited 2019 February 24, 2019]. Available from: http://gco.iarc.fr/today/fact-sheets-populations
Sandler S. Esophagogastric junction and gastric adenocarcinoma: neoadjuvant and adjuvant therapy, and future directions. Oncology (Williston Park). 2014;28:505-12.
Nebot-Bral L, Brandao D, Verlingue L, Rouleau E, Caron O, Despras E, et al. Hypermutated tumors in the era of immunotherapy: The paradigm of personalized medicine. Eur J Cancer. 2017;84:290-303. https://doi.org/10.1016/j.ejca.2017.07.026
Cancer Genome Atlas Research N. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513(7517):202-9. https://doi.org/10.1038/nature13480
Ratti M, Lampis A, Hahne JC, Passalacqua R, Valeri N. Microsatellite instability in gastric cancer: molecular bases, clinical perspectives, and new treatment approaches. Cell Mol Life Sci. 2018;75:4151-62. https://doi.org/10.1007/s00018-018-2906-9
Fuchs CS, Doi T, Jang RW, Muro K, Satoh T, Machado M, et al. Safety and Efficacy of Pembrolizumab Monotherapy in Patients with Previously Treated Advanced Gastric and Gastroesophageal Junction Cancer: Phase 2 Clinical KEYNOTE-059 Trial. JAMA Oncol. 2018;4:e180013.
Cárdenas W, Castillo A, Vargas C, Moreno O, Insuasti J. Análisis de la inestabilidad de microsatélites mediante el marcador BAT-26 en una muestra de pacientes del Hospital Universitario de Santander con diagnóstico de cáncer gástrico o colorrectal. Colombia Médica. 2008;39(Supl.2):41-51.
Sepulveda AR, Santos AC, Yamaoka Y, Wu L, Gutierrez O, Kim JG, et al. Marked differences in the frequency of microsatellite instability in gastric cancer from different countries. Am J Gastroenterol. 1999;94:3034-8. https://doi.org/10.1111/j.1572-0241.1999.01453.x
Piñeros M, Pardo C, Gamboa O, Hernández G. Atlas de Mortalidad por Cáncer en Colombia. Bogotá: Instituto Nacional de Cancerología-Instituto Geográfico Agustín Codazzi; 2010.
Ashraf N, Hoffe S, Kim R. Adjuvant treatment for gastric cancer: chemotherapy versus radiation. Oncologist. 2013;18:1013-21. https://doi.org/10.1634/theoncologist.2012-0462
Macdonald JS, Smalley SR, Benedetti J, Hundahl SA, Estes NC, Stemmermann GN, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001;345:725-30. https://doi.org/10.1056/NEJMoa010187
Smalley SR, Benedetti JK, Haller DG, Hundahl SA, Estes NC, Ajani JA, et al. Updated analysis of SWOG-directed intergroup study 0116: a phase III trial of adjuvant radiochemotherapy versus observation after curative gastric cancer resection. J Clin Oncol. 2012;30:2327-33. https://doi.org/10.1200/JCO.2011.36.7136
Soon YY, Leong CN, Tey JC, Tham IW, Lu JJ. Postoperative chemo-radiotherapy versus chemotherapy for resected gastric cancer: a systematic review and meta-analysis. J Med Imag Radiat Oncol. 2014;58:483-96. https://doi.org/10.1111/1754-9485.12190
Boige V, Pignon J, Saint-Aubert B, Lasser P, Conroy T, Bouché O, et al. Final results of a randomized trial comparing preoperative 5-fluorouracil (F)/cisplatin (P) to surgery alone in adenocarcinoma of stomach and lower esophagus (ASLE): FNLCC ACCORD07-FFCD 9703 trial. J Clin Oncol. 2007;25(18_suppl):4510.
Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJ, Nicolson M, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355:11-20. https://doi.org/10.1056/NEJMoa055531
Schuhmacher C, Gretschel S, Lordick F, Reichardt P, Hohenberger W, Eisenberger CF, et al. Neoadjuvant chemotherapy compared with surgery alone for locally advanced cancer of the stomach and cardia: European Organisation for Research and Treatment of Cancer randomized trial 40954. J Clin Oncol. 2010;28:5210-8. https://doi.org/10.1200/JCO.2009.26.6114
Ychou M, Boige V, Pignon JP, Conroy T, Bouche O, Lebreton G, et al. Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter phase III trial. J Clin Oncol. 2011;29:1715-21. https://doi.org/10.1200/JCO.2010.33.0597
Cascinu S, Labianca R, Barone C, Santoro A, Carnaghi C, Cassano A, et al. Adjuvant treatment of high-risk, radically resected gastric cancer patients with 5-fluorouracil, leucovorin, cisplatin, and epidoxorubicin in a randomized controlled trial. J Natl Cancer Inst. 2007;99:601-7. https://doi.org/10.1093/jnci/djk131
Drake JW, Charlesworth B, Charlesworth D, Crow JF. Rates of spontaneous mutation. Genetics. 1998;148:1667-86.
Kunkel TA. DNA replication fidelity. J Biol Chem. 2004;279:16895-8. https://doi.org/10.1074/jbc.R400006200
Ruiz-Banobre J, Goel A. DNA Mismatch Repair Deficiency and Immune Checkpoint Inhibitors in Gastrointestinal Cancers. Gastroenterology. 2019;156:890-903. https://doi.org/10.1053/j.gastro.2018.11.071
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646-74. https://doi.org/10.1016/j.cell.2011.02.013
Dietlein F, Thelen L, Reinhardt HC. Cancer-specific defects in DNA repair pathways as targets for personalized therapeutic approaches. Trends Genet. 2014;30:326-39. https://doi.org/10.1016/j.tig.2014.06.003
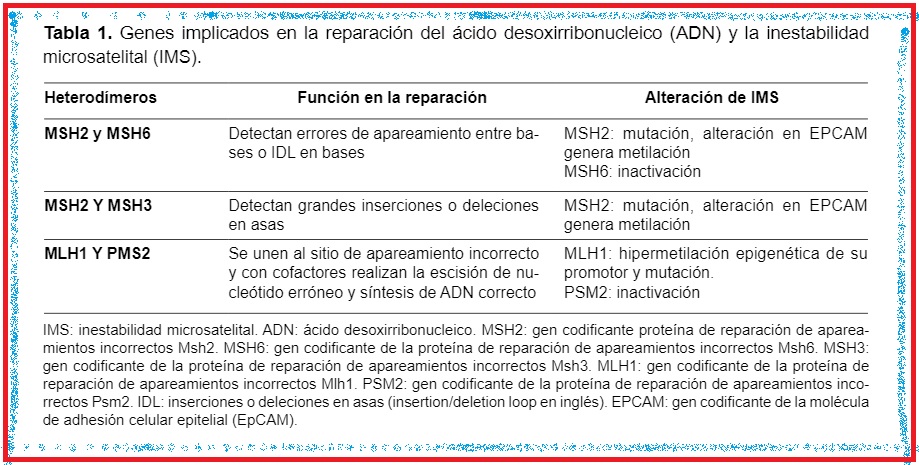
Genschel J, Littman SJ, Drummond JT, Modrich P. Isolation of MutSbeta from human cells and comparison of the mismatch repair specificities of MutSbeta and MutSalpha. J Biol Chem. 1998;273:19895-901. https://doi.org/10.1074/jbc.273.31.19895
Cortes-Ciriano I, Lee S, Park WY, Kim TM, Park PJ. A molecular portrait of microsatellite instability across multiple cancers. Nature communications. 2017;8:151-8. https://doi.org/10.1038/ncomms15180
Muller MF, Ibrahim AE, Arends MJ. Molecular pathological classification of colorectal cancer. Virchows Archiv. 2016;469:125-34. https://doi.org/10.1007/s00428-016-1956-3
Jiricny J. The multifaceted mismatch-repair system. Nat Rev Mol Cell Biol. 2006;7:335-46. https://doi.org/10.1038/nrm1907
Kovacs ME, Papp J, Szentirmay Z, Otto S, Olah E. Deletions removing the last exon of TACSTD1 constitute a distinct class of mutations predisposing to Lynch syndrome. Human mutation. 2009;30:197-203. https://doi.org/10.1002/humu.20942
Lynch HT, Shaw MW, Magnuson CW, Larsen AL, Krush AJ. Hereditary factors in cancer. Study of two large midwestern kindreds. Arch Inter Med. 1966;117:206-12. https://doi.org/10.1001/archinte.1966.03870080050009
Boland CR, Troncale FJ. Familial colonic cancer without antecedent polyposis. Ann Inter Med. 1984;100:700-1. https://doi.org/10.7326/0003-4819-100-5-700
Giardiello FM, Allen JI, Axilbund JE, Boland CR, Burke CA, Burt RW, et al. Guidelines on genetic evaluation and management of Lynch syndrome: a consensus statement by the US Multi-Society Task Force on colorectal cancer. Gastroenterology. 2014;147:502-26. https://doi.org/10.1053/j.gastro.2014.04.001
Knudson AG, Jr. Hereditary cancer, oncogenes, and antioncogenes. Cancer Research. 1985;45:1437-43.
Hendriks YM, Wagner A, Morreau H, Menko F, Stormorken A, Quehenberger F, et al. Cancer risk in hereditary nonpolyposis colorectal cancer due to MSH6 mutations: impact on counseling and surveillance. Gastroenterology. 2004;127(1):17-25. https://doi.org/10.1053/j.gastro.2004.03.068
Senter L, Clendenning M, Sotamaa K, Hampel H, Green J, Potter JD, et al. The clinical phenotype of Lynch syndrome due to germ-line PMS2 mutations. Gastroenterology. 2008;135(2):419-28. https://doi.org/10.1053/j.gastro.2008.04.026
Edelstein DL, Axilbund J, Baxter M, Hylind LM, Romans K, Griffin CA, et al. Rapid development of colorectal neoplasia in patients with Lynch syndrome. Clin Gastroenterol Hepatol. 2011;9:340-3. https://doi.org/10.1016/j.cgh.2010.10.033
Quehenberger F, Vasen HF, van Houwelingen HC. Risk of colorectal and endometrial cancer for carriers of mutations of the hMLH1 and hMSH2 gene: correction for ascertainment. J Med Genet. 2005;42:491-6. https://doi.org/10.1136/jmg.2004.024299
Lynch HT, Lynch PM, Pester J, Fusaro RM. The cancer family syndrome. Rare cutaneous phenotypic linkage of Torre's syndrome. Arch Inter Med. 1981;141:607-11. https://doi.org/10.1001/archinte.1981.00340050059016
Kastrinos F, Stoffel EM, Balmana J, Steyerberg EW, Mercado R, Syngal S. Phenotype comparison of MLH1 and MSH2 mutation carriers in a cohort of 1,914 individuals undergoing clinical genetic testing in the United States. Cancer Epidemiol Biomarkers Prev. 2008;17:2044-51. https://doi.org/10.1158/1055-9965.EPI-08-0301
Entius MM, Keller JJ, Drillenburg P, Kuypers KC, Giardiello FM, Offerhaus GJ. Microsatellite instability and expression of hMLH-1 and hMSH-2 in sebaceous gland carcinomas as markers for Muir-Torre syndrome. Clinical Cancer. 2000;6:1784-9.
Durno CA, Holter S, Sherman PM, Gallinger S. The gastrointestinal phenotype of germline biallelic mismatch repair gene mutations. Am J Gastroenterol. 2010;105:2449-56. https://doi.org/10.1038/ajg.2010.215
Trimbath JD, Petersen GM, Erdman SH, Ferre M, Luce MC, Giardiello FM. Cafe-au-lait spots and early onset colorectal neoplasia: a variant of HNPCC? Fam Cancer. 2001;1:101-5.
Berg KD, Glaser CL, Thompson RE, Hamilton SR, Griffin CA, Eshleman JR. Detection of microsatellite instability by fluorescence multiplex polymerase chain reaction. J Mol Diagn. 2000;2(1):20-8. https://doi.org/10.1016/S1525-1578(10)60611-3
Murphy KM, Zhang S, Geiger T, Hafez MJ, Bacher J, Berg KD, et al. Comparison of the microsatellite instability analysis system and the Bethesda panel for the determination of microsatellite instability in colorectal cancers. J Mol Diagn. 2006;8(3):305-11. https://doi.org/10.2353/jmoldx.2006.050092
Funkhouser WK, Jr., Lubin IM, Monzon FA, Zehnbauer BA, Evans JP, Ogino S, et al. Relevance, pathogenesis, and testing algorithm for mismatch repair-defective colorectal carcinomas: a report of the association for molecular pathology. J Mol Diagn. 2012;14(2):91-103. https://doi.org/10.1016/j.jmoldx.2011.11.001
Leite M, Corso G, Sousa S, Milanezi F, Afonso LP, Henrique R, et al. MSI phenotype and MMR alterations in familial and sporadic gastric cancer. Int J Cancer. 2011;128:1606-13. https://doi.org/10.1002/ijc.25495
Vaccaro CA, Carrozzo JE, Mocetti E, Berho M, Valdemoros P, Mullen E, et al. Expresión inmunohistoquímica e inestabilidad microsatelital en el síndrome de Lynch. Medicina (Buenos Aires). 2007;67:274-8.
Kulangara K, Zhang N, Corigliano E, Guerrero L, Waldroup S, Jaiswal D, et al. Clinical Utility of the Combined Positive Score for Programmed Death Ligand-1 Expression and the Approval of Pembrolizumab for Treatment of Gastric Cancer. Arch Pathol Lab Med. 2019;143:330-7. https://doi.org/10.5858/arpa.2018-0043-OA
Chen Q, Li T, Yue W. Drug response to PD-1/PD-L1 blockade: based on biomarkers. OncoTargets Ther. 2018;11:4673-83. https://doi.org/10.2147/OTT.S168313
Baumeister SH, Freeman GJ, Dranoff G, Sharpe AH. Coinhibitory Pathways in Immunotherapy for Cancer. Annual review of immunology. 2016;34:539-73. https://doi.org/10.1146/annurev-immunol-032414-112049
Overman MJ, McDermott R, Leach JL, Lonardi S, Lenz HJ, Morse MA, et al. Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): an open-label, multicentre, phase 2 study. The Lancet Oncology. 2017;18:1182-91. https://doi.org/10.1016/S1470-2045(17)30422-9
Overman MJ, Lonardi S, Wong KYM, Lenz HJ, Gelsomino F, Aglietta M, et al. Durable clinical benefit with nivolumab plus ipilimumab in DNA mismatch repair-deficient/microsatellite instability-high metastatic colorectal cancer. J Clin Oncol. 2018;36:773-9. https://doi.org/10.1200/JCO.2017.76.9901
Lin EM, Gong J, Klempner SJ, Chao J. Advances in immuno-oncology biomarkers for gqstroesophageal cancer: Programmed death ligand 1, microsatellite instability, and beyond. World J Gastroenterol. 2018;24:2686-97. https://doi.org/10.3748/wjg.v24.i25.2686
Smyth EC, Wotherspoon A, Peckitt C, Gonzalez D, Hulkki-Wilson S, Eltahir Z, et al. Mismatch Repair Deficiency, Microsatellite Instability, and Survival: An Exploratory Analysis of the Medical Research Council Adjuvant Gastric Infusional Chemotherapy (MAGIC) Trial. JAMA Oncol. 2017;3:1197-203. https://doi.org/10.1001/jamaoncol.2016.6762
Polom K, Marano L, Marrelli D, De Luca R, Roviello G, Savelli V, et al. Meta-analysis of microsatellite instability in relation to clinicopathological characteristics and overall survival in gastric cancer. Br J Surg. 2018;105:159-67.n https://doi.org/10.1002/bjs.10663
Publicado
Cómo citar
Número
Sección
Licencia
Todos los textos incluidos en la Revista Colombiana de Cirugía están protegidos por derechos de autor. Las opiniones expresadas en los artículos firmados son las de los autores y no coinciden necesariamente con las de los directores o los editores de la Revista Colombiana de Cirugía. Las sugerencias diagnósticas o terapéuticas como elección de productos, dosificación y métodos de empleo corresponden a la experiencia y al criterio de los autores.

















.jpg)